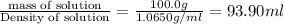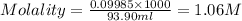Chemistry, 13.02.2020 21:43 PONBallfordM89
What is the molarity and molality of a solution that is 10.00 % by mass potassium hydrogen carbonate (KHCO3, 100.11 g/mol) and has a density of 1.0650 g/mL

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3


Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
What is the molarity and molality of a solution that is 10.00 % by mass potassium hydrogen carbonate...
Questions

Physics, 19.10.2019 10:30

Mathematics, 19.10.2019 10:30




Mathematics, 19.10.2019 10:30

English, 19.10.2019 10:30




History, 19.10.2019 10:30


Computers and Technology, 19.10.2019 10:30

Mathematics, 19.10.2019 10:30

Mathematics, 19.10.2019 10:30

Mathematics, 19.10.2019 10:30


German, 19.10.2019 10:30

Mathematics, 19.10.2019 10:30

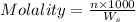
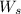 = weight of solvent in g
= weight of solvent in g 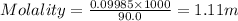
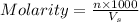
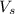 = volume of solution in ml
= volume of solution in ml