
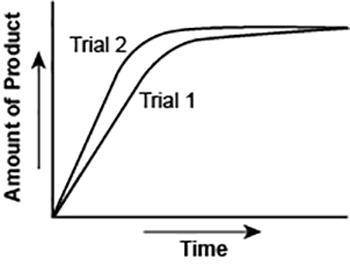
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different concentration of reactant was used during each trial, whereas the other factors were kept constant.
Which of the following statements explains which trial has a *lower* concentration of the reactant? (5 points)
Trial 1, because the average rate of the reaction is lower.
Trial 1, because this reaction lasted for a longer duration than Trial 2.
Trial 2, because this reaction was initially fast and later slowed down.
Trial 2, because the volume of product formed per unit time was higher.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1


Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different...
Questions

Mathematics, 23.07.2019 05:20


Mathematics, 23.07.2019 05:20

Mathematics, 23.07.2019 05:20

Mathematics, 23.07.2019 05:20


Mathematics, 23.07.2019 05:20

Mathematics, 23.07.2019 05:20


Mathematics, 23.07.2019 05:20

Mathematics, 23.07.2019 05:20


Mathematics, 23.07.2019 05:20

Mathematics, 23.07.2019 05:20


Mathematics, 23.07.2019 05:20

Mathematics, 23.07.2019 05:20

Biology, 23.07.2019 05:20


Mathematics, 23.07.2019 05:20



