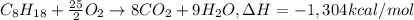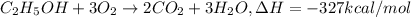Chemistry, 14.02.2020 02:02 blackjack73
Ethanol is added to gasoline to produce E-15 and E-85. It promotes more complete combustion of the gasoline and is an octane booster. Compare the heats of combustion of 2,2,4-trimethylpentane (1304 kcal/mol) and ethanol (327 kcal/mol). Which has the higher heat of combustion in kcal/mol?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


Chemistry, 22.06.2019 22:10
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3

Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2
You know the right answer?
Ethanol is added to gasoline to produce E-15 and E-85. It promotes more complete combustion of the g...
Questions



Mathematics, 17.10.2020 06:01


Mathematics, 17.10.2020 06:01

History, 17.10.2020 06:01

Mathematics, 17.10.2020 06:01


Mathematics, 17.10.2020 06:01


Advanced Placement (AP), 17.10.2020 06:01

Mathematics, 17.10.2020 06:01

Computers and Technology, 17.10.2020 06:01


Mathematics, 17.10.2020 06:01

Health, 17.10.2020 06:01




Computers and Technology, 17.10.2020 06:01