Chemistry, 14.02.2020 03:01 cordovatierra16
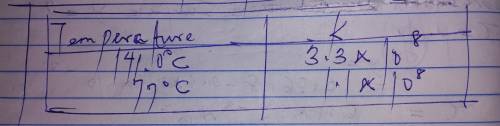
The rate constant for a certain reaction is measured at two different temperatures: temperature Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy for this reaction. Round your answer to significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2
You know the right answer?
The rate constant for a certain reaction is measured at two different temperatures: temperature Assu...
Questions


Advanced Placement (AP), 10.04.2020 10:58

History, 10.04.2020 10:58


Physics, 10.04.2020 10:58

Mathematics, 10.04.2020 10:59





Physics, 10.04.2020 10:59



English, 10.04.2020 10:59

Mathematics, 10.04.2020 10:59

Health, 10.04.2020 10:59

Chemistry, 10.04.2020 10:59



Mathematics, 10.04.2020 11:00