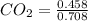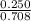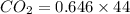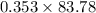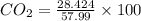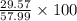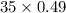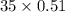A mixture of CO2 and Kr weighs 35.0 g and exerts a pressure of 0.708 atm in its container. Since Kr is expensive, you wish to recover it from the mixture. After the CO2 is completely removed by absorption with NaOH(s), the pressure in the container is 0.250 atm. How many grams of CO2 were originally present? How many grams of Kr can you recover?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:10
The rock in a lead ore deposit contains 89 % pbs by mass. how many kilograms of the rock must be processed to obtain 1.5 kg of pb?
Answers: 1

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2
You know the right answer?
A mixture of CO2 and Kr weighs 35.0 g and exerts a pressure of 0.708 atm in its container. Since Kr...
Questions




History, 30.08.2019 00:30

Mathematics, 30.08.2019 00:30


Mathematics, 30.08.2019 00:30






Mathematics, 30.08.2019 00:30




English, 30.08.2019 00:30



History, 30.08.2019 00:30

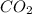 will be calculated as follows.
will be calculated as follows.