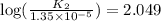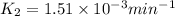Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1
You know the right answer?
The gas phase reaction 2 N2O5(g) → 4 NO2(g) + O2(g) has an activation energy of 103 kJ/mol, and the...
Questions

Mathematics, 04.02.2020 09:45



Mathematics, 04.02.2020 09:45


Geography, 04.02.2020 09:45






Mathematics, 04.02.2020 09:45

Chemistry, 04.02.2020 09:45

Mathematics, 04.02.2020 09:45


Mathematics, 04.02.2020 09:45


Mathematics, 04.02.2020 09:45


Chemistry, 04.02.2020 09:45

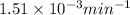
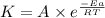
![\log (\frac{K_2}{K_1})=\frac{Ea}{2.303\times R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0511/7907/6d953.png)
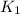 = rate constant at 266 K =
= rate constant at 266 K = 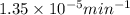
 = rate constant at 296 K = ?
= rate constant at 296 K = ?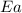 = activation energy for the reaction = 103 kJ/mol = 103000 J/mol
= activation energy for the reaction = 103 kJ/mol = 103000 J/mol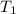 = initial temperature = 266 K
= initial temperature = 266 K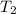 = final temperature = 296 K
= final temperature = 296 K![\log (\frac{K_2}{1.35\times 10^{-5}})=\frac{103000}{2.303\times 8.314J/mole.K}[\frac{1}{266}-\frac{1}{296}]](/tpl/images/0511/7907/d42c0.png)