Chemistry, 15.02.2020 02:59 Nyasiahenry
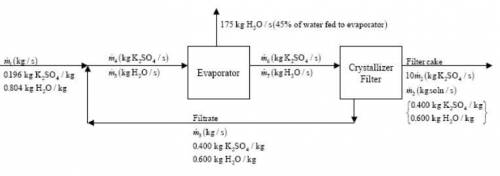
An evaporation–crystallization process of the type described in Example 4.5-2 is used to obtain solid potassium sulfate from an aqueous solution of this salt. The fresh feed to the process contains 19.6 wt% K2SO4. The wet filter cake consists of solid K2SO4 crystals and a 40.0 wt% K2SO4 solution, in a ratio 10 kg crystals/kg solution. The filtrate, also a 40.0% solution, is recycled to join the fresh feed. Of the water fed to the evaporator, 45.0% is evaporated. The evaporator has a maximum capacity of 175 kg water evaporated/s. Calculate the maximum production rate of solid K2SO4, the rate at which fresh feed must be supplied to achieve this production rate.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Construct the hypothetical phase diagram for metals a and b between room temperature (20c) and 700c, given the following information: * the melting temperature of metal a is 480c. • the maximum solubility of b in a is 4 wt% b, which occurs at 420c. • the solubility of b in a at room temperature is 0 wt% b. • one eutectic occurs at 420c and 18 wt% b–82 wt% a. • a second eutectic occurs at 475c and 42 wt% b–58 wt% a. • the intermetallic compound ab exists at a composition of 30 wt% b–70 wt% a, and melts congruently at 525c.• the melting temperature of metal b is 600c. • the maximum solubility of a in b is 13 wt% a, which occurs at 475c. • the solubility of a in b at room temperature is 3 wt% a.
Answers: 1

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

Chemistry, 22.06.2019 16:00
Uranium can supply energy for the worlds electricity without admitting harmful greenhouse gases which of these statements best describes an outcome of uranium mining
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3
You know the right answer?
An evaporation–crystallization process of the type described in Example 4.5-2 is used to obtain soli...
Questions










Mathematics, 26.05.2021 18:50


Mathematics, 26.05.2021 18:50



Computers and Technology, 26.05.2021 18:50



Mathematics, 26.05.2021 18:50

History, 26.05.2021 18:50

Mathematics, 26.05.2021 18:50

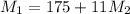
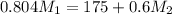
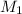 = 220.77 kg/s
= 220.77 kg/s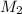 = 4.16 kg/s
= 4.16 kg/s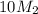 = 10*4.16 = 416 kg/s
= 10*4.16 = 416 kg/s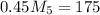
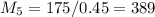 kg/s
kg/s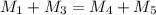
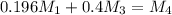
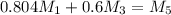
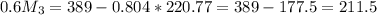
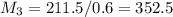 kg/s
kg/s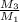 = 352.5/220.77 = 1.6
= 352.5/220.77 = 1.6