
Chemistry, 17.02.2020 17:26 sedilei1515
Two equilibrium reactions of nitrogen with oxygen, with their corresponding equilibrium constants (Kc) at a certain temperature, are given below. reaction (1): N2(g) + O2(g) 2 NO(g); Kc = 1.54e-31 reaction (2): N2(g) + 1/2 O2(g) N2O(g); Kc = 2.61e-24 Using this set of data, determine the equilibrium constant for the following reaction, at the same temperature. reaction (3): N2O(g) + 1/2 O2(g) 2 NO(g)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
The crust of earth may a- continets and ocean floors. b-continents only. c-layers of sedimentary rocks and continents. d-all of the above
Answers: 2

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1
You know the right answer?
Two equilibrium reactions of nitrogen with oxygen, with their corresponding equilibrium constants (K...
Questions



Mathematics, 27.02.2021 23:40

History, 27.02.2021 23:40






Mathematics, 27.02.2021 23:40

History, 27.02.2021 23:40

History, 27.02.2021 23:40


Health, 27.02.2021 23:40



Mathematics, 27.02.2021 23:40



English, 27.02.2021 23:40

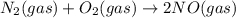 ; Kc = 1.54e - 31
; Kc = 1.54e - 31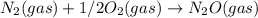 ; Kc = 2.16e - 24
; Kc = 2.16e - 24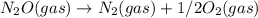 Kc = 1/2.16e - 24
Kc = 1/2.16e - 24 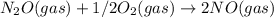 Kc = 1.54e-31 × 1/2.61e - 24
Kc = 1.54e-31 × 1/2.61e - 24

