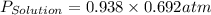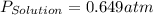Chemistry, 17.02.2020 17:57 Alienhead6187
12. The vapor pressure of water at 90°C is 0.692 atm. What is the vapor pressure (in atm) of a solution made by dissolving 3.68 mole(s) of CsF(s) in 1.00 kg of water? Assume that Raoult's law applies.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1


Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1
You know the right answer?
12. The vapor pressure of water at 90°C is 0.692 atm. What is the vapor pressure (in atm) of a solut...
Questions





History, 17.01.2020 21:31

Mathematics, 17.01.2020 21:31

Biology, 17.01.2020 21:31

Social Studies, 17.01.2020 21:31


English, 17.01.2020 21:31


History, 17.01.2020 21:31

Computers and Technology, 17.01.2020 21:31



Mathematics, 17.01.2020 21:31



English, 17.01.2020 21:31

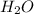 = 1.00 kg = 1000 g
= 1.00 kg = 1000 g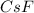 = 3.68 mole
= 3.68 mole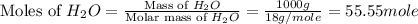
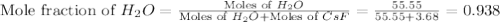
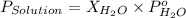
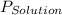 = vapor pressure of solution
= vapor pressure of solution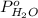 = vapor pressure of water = 0.692 atm
= vapor pressure of water = 0.692 atm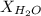 = mole fraction of water = 0.938
= mole fraction of water = 0.938