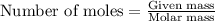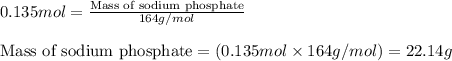Chemistry, 17.02.2020 22:18 kayla114035
Hard water often contains dissolved Ca2 and Mg2 ions. One way to soften water is to add phosphates. The phosphate ion forms insoluble precipitates with calcium and magnesium ions, removing them from solution. A solution is 0.050 M in calcium chloride and 0.085 M in magnesium nitrate. What mass of sodium phosphate would you add to 1.5 L of this solution to completely eliminate the hard water ions.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 21:00
Which of these is an example of pseudoscience? a) predicting the time of sunrise based on data on position of earth b) predicting the date of the moon phases based on data on position of earth c) predicting eclipses based on the position of the sun and the moon d) predicting future events in a person's life based on the position of the moon
Answers: 1
You know the right answer?
Hard water often contains dissolved Ca2 and Mg2 ions. One way to soften water is to add phosphates....
Questions

Mathematics, 17.09.2019 15:30








Social Studies, 17.09.2019 15:30

Health, 17.09.2019 15:30

Computers and Technology, 17.09.2019 15:30

History, 17.09.2019 15:30

Spanish, 17.09.2019 15:30


Mathematics, 17.09.2019 15:30


Mathematics, 17.09.2019 15:30


Mathematics, 17.09.2019 15:30

English, 17.09.2019 15:30

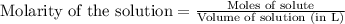 .....(1)
.....(1)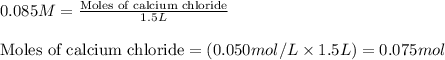
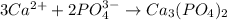
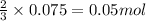 of phosphate ions
of phosphate ions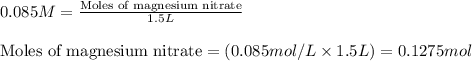
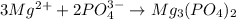
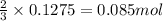 of phosphate ions
of phosphate ions