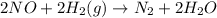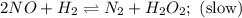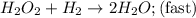Chemistry, 17.02.2020 23:38 cordobamariana07
Nitrogen monoxide, NO, reacts with hydrogen, H2, according to the following equation. 2 NO + 2 H2 → N2 + 2 H2O What would the rate law be if the mechanism for this reaction were as follows? (Rate expressions take the general form: rate = k . [A]a . [B]b.) 2 NO + H2 → N2 + H2O2 (slow) H2O2 + H2 → 2 H2O (fast)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Long term exposure to waves can cause sunburns and skin cancer. a) visible b) infrared c) gamma rays d) ultraviole
Answers: 1

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2

Chemistry, 23.06.2019 07:10
Which one of the following is an oxidation-reduction reaction? naoh + hno3 --> h2o + kno3 naoh + hno3 --> h2o + kno3 so3 + h2o --> h2so4 cacl2 + na2co3 --> caco3 + 2 nacl ch4 + 2 o2 --> co2 + 2 h2o al2(so4)3 + 6 koh --> 2 al(oh)3 + 3 k2so4
Answers: 3
You know the right answer?
Nitrogen monoxide, NO, reacts with hydrogen, H2, according to the following equation. 2 NO + 2 H2 →...
Questions


Chemistry, 27.04.2021 06:50



Mathematics, 27.04.2021 06:50

Geography, 27.04.2021 06:50

Mathematics, 27.04.2021 06:50


Mathematics, 27.04.2021 06:50

Mathematics, 27.04.2021 06:50





Mathematics, 27.04.2021 06:50



SAT, 27.04.2021 06:50


![\text{Rate}=k[NO]^2[H_2]](/tpl/images/0513/5186/3b203.png)