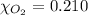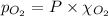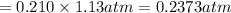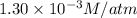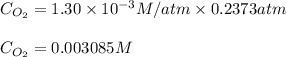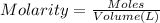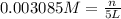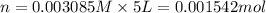Chemistry, 18.02.2020 03:26 netflixacc0107
Calculate the mass of oxygen gas (O2) dissolved in a 5.00 L bucket of water exposed to a pressure of 1.13 atm of air. Assume the mole fraction of oxygen in air to be 0.210 and the Henry's law constant for air in water at this temperature to be 1.30 × 10-3 M/atm. Report your answer in mg.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asmall amount of a solid is added to water. the observation made after fifteen minutes is shown in the figure. which of these solids has been probably added to water? a) oil b) sand c) sugar d) wood chips
Answers: 1

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2

You know the right answer?
Calculate the mass of oxygen gas (O2) dissolved in a 5.00 L bucket of water exposed to a pressure of...
Questions



Spanish, 14.07.2019 12:30


History, 14.07.2019 12:30




Computers and Technology, 14.07.2019 12:30

Physics, 14.07.2019 12:30



Mathematics, 14.07.2019 12:30


Chemistry, 14.07.2019 12:30



Mathematics, 14.07.2019 12:30

Mathematics, 14.07.2019 12:30

Biology, 14.07.2019 12:30

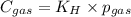
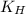 = Henry's constant =
= Henry's constant = 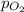 = partial pressure of oxygen
= partial pressure of oxygen