
Chemistry, 18.02.2020 03:29 onewaydemon
The iodide ion reacts with hypochlorite ion (the active ingredient in chlorine bleaches) in the following way:
OCl- + I- → OI-1 +Cl-.
This rapid reaction gives the following rate data:
[OCl-](M) [I]- (M) Rate (M/s)
1.5×10^3 1.5×10^3 1.36×10^4
3.0×10^3 1.5×10^3 2.72×10^4
1.5×10^3 3.0×10^3 2.72×10^4
Write the rate law for this reaction.
Calculate the rate constant with proper units.
Calculate the rate when [OCl-]= 1.8×10^3 M and [I-]= 6.0×10^4 M .

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
What three natural resources are found in the great lakes region
Answers: 2

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 23.06.2019 03:00
What do electromagnetic waves carry? how are they produced through which media can they move? where do they transfer energy? what do they not transfer? what do mechanical waves carry? how are they produced? through which media can they move? where do they transfer energy? what do they not transfer?
Answers: 1

Chemistry, 23.06.2019 04:20
The lewis diagrams for magnesium and fluorine are shown below. what is the correct chemical formula for magnesium fluoride? a. mgf b. mg2f c. mgf2 d. mg2f3
Answers: 1
You know the right answer?
The iodide ion reacts with hypochlorite ion (the active ingredient in chlorine bleaches) in the foll...
Questions



Social Studies, 26.01.2020 15:31


History, 26.01.2020 15:31

English, 26.01.2020 15:31





Arts, 26.01.2020 15:31

Chemistry, 26.01.2020 15:31

Mathematics, 26.01.2020 15:31

English, 26.01.2020 15:31

World Languages, 26.01.2020 15:31

Mathematics, 26.01.2020 15:31

Mathematics, 26.01.2020 15:31

Mathematics, 26.01.2020 15:31

Mathematics, 26.01.2020 15:31

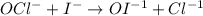
![K \times [OCl^{-}] \times [l^{-}]](/tpl/images/0513/8261/88013.png)
![K [OCl^{-}][l^{-}]](/tpl/images/0513/8261/94fd7.png)
![[OCl^{-}] = [l^{-}]](/tpl/images/0513/8261/5e8f1.png) )
)
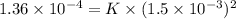
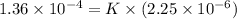
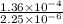
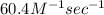
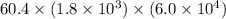
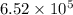
![[OCl^{-}] = 1.8 \times 10^{3}](/tpl/images/0513/8261/c42db.png) M and
M and ![[I^{-}]= 6.0 \times 10^{4}](/tpl/images/0513/8261/40481.png) M is
M is 

