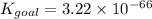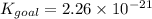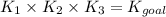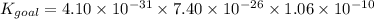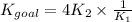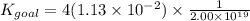Chemistry, 18.02.2020 23:57 briannaleiigh
Determine the value of the equilibrium constant, Kgoal, for the reaction
N2(g)+H2O(g)⇌NO(g)+12N2H4(g), Kgoal=?
by making use of the following information:
1. N2(g)+O2(g)⇌2NO(g), K1 = 4.10×10−31
2. N2(g)+2H2(g)⇌N2H4(g), K2 = 7.40×10−26
3. 2H2O(g)⇌2H2(g)+O2(g), K3 = 1.06×10−10
Express your answer numerically.
Kgoal =
Part B
Determine the equilibrium constant, Kgoal, for the reaction
4PCl5(g)⇌P4(s)+10Cl2(g), Kgoal=?
by making use of the following information:
P4(s)+6Cl2(g)⇌4PCl3(g), K1=2.00×1019
PCl5(g)⇌PCl3(g)+Cl2(g), K2=1.13×10−2
Express your answer numerically.
Kgoal =

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 00:30
What must happen before a body cell can begin mitotic cell division
Answers: 2


You know the right answer?
Determine the value of the equilibrium constant, Kgoal, for the reaction
N2(g)+H2O(g)⇌NO...
N2(g)+H2O(g)⇌NO...
Questions



Mathematics, 14.12.2019 04:31




Computers and Technology, 14.12.2019 04:31