
Chemistry, 19.02.2020 00:21 carlyfaith3375
When 60 mL of 1.30 mol/L AgNO3(aq) and 60 mL of 1.30 mol/L HCl(aq) are mixed in a simple calorimeter, the temperature rises by 5.18°C. The molar enthalpy of reaction of HCl(aq) is ab. C kJ/mol.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3
You know the right answer?
When 60 mL of 1.30 mol/L AgNO3(aq) and 60 mL of 1.30 mol/L HCl(aq) are mixed in a simple calorimeter...
Questions

Biology, 03.10.2019 10:50

Mathematics, 03.10.2019 10:50






English, 03.10.2019 10:50

Spanish, 03.10.2019 10:50

Biology, 03.10.2019 10:50

Biology, 03.10.2019 10:50


English, 03.10.2019 10:50

Mathematics, 03.10.2019 10:50

Spanish, 03.10.2019 10:50





Mathematics, 03.10.2019 10:50

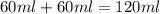
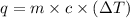
 = specific heat of water =
= specific heat of water = 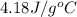
 = change in temperature =
= change in temperature = 
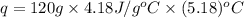

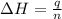
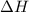 = enthalpy of neutralization = ?
= enthalpy of neutralization = ?


