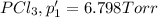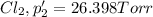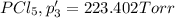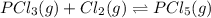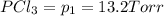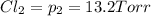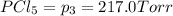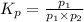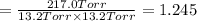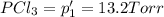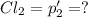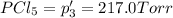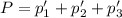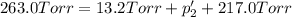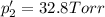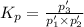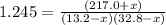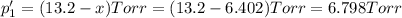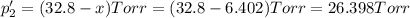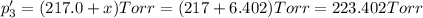An equilibrium mixture of PCl5(g), PCl3(g), and Cl2(g) has partial pressures of 217.0 Torr, 13.2 Torr, and 13.2 Torr, respectively. A quantity of Cl2(g) is injected into the mixture, and the total pressure jumps to 263.0 Torr (at the moment of mixing). The system then re-equilibrates. The appropriate chemical equation is PCl3(g)+Cl2(g) <-->PCl5(g) Calculate the new partial pressures after equilibrium is reestablished in torr

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1
You know the right answer?
An equilibrium mixture of PCl5(g), PCl3(g), and Cl2(g) has partial pressures of 217.0 Torr, 13.2 Tor...
Questions

History, 16.10.2019 19:10

Mathematics, 16.10.2019 19:10

Mathematics, 16.10.2019 19:10

Mathematics, 16.10.2019 19:10




History, 16.10.2019 19:10

Mathematics, 16.10.2019 19:10



Chemistry, 16.10.2019 19:10








Chemistry, 16.10.2019 19:10