
Chemistry, 19.02.2020 05:52 animerocks07
Determining reaction order : Rate Laws(Chemistry)
The reaction of nitric oxide with hydrogen at 1280C is as follows:
2NO(g) +2H2 (g) -> N2(g) + 2H2O(g)
From the following experimental data, determine the rate law and the rate constant.
30 POINTS!


Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
How much energy moves onto the next level, in an energy pyramid
Answers: 1

Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1
You know the right answer?
Determining reaction order : Rate Laws(Chemistry)
The reaction of nitric oxide with hydrogen...
The reaction of nitric oxide with hydrogen...
Questions




Mathematics, 17.11.2020 02:10



Mathematics, 17.11.2020 02:10

Computers and Technology, 17.11.2020 02:10

Spanish, 17.11.2020 02:10

Business, 17.11.2020 02:10


French, 17.11.2020 02:10

Physics, 17.11.2020 02:10



Health, 17.11.2020 02:10


Mathematics, 17.11.2020 02:10


English, 17.11.2020 02:10

![R=k[NO]3[H_2]^1](/tpl/images/0515/4935/da8f4.png)
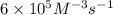
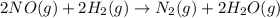
 in rate law be x and y .
in rate law be x and y .![R=k[NO]^[H_2]^y](/tpl/images/0515/4935/cf38a.png)
![[NO]=0.0100 M, [H_2]=0.0100 M](/tpl/images/0515/4935/aa668.png)
![0.00600 M/s=k[0.0100 M]^x[0.0100 M]^y](/tpl/images/0515/4935/a6007.png) ..[1]
..[1]![[NO]=0.0200 M, [H_2]=0.0300 M](/tpl/images/0515/4935/ba703.png)
![0.144 M/s=k[0.0200 M]^x[0.0300 M]^y](/tpl/images/0515/4935/0042d.png) ..[2]
..[2]![[NO]=0.0100 M, [H_2]=0.0200 M](/tpl/images/0515/4935/ba63c.png)
![0.0120 M/s=k[0.0100 M]^x[0.0200 M]^y](/tpl/images/0515/4935/9f0d9.png) ..[3]
..[3]![\frac{0.00600 M/s}{0.0120 M/s}=\frac{k[0.0100 M]^x\times [0.0100 M]^y}{k[0.0100 M]^x\times [0.0200 M]^y}](/tpl/images/0515/4935/1b196.png)
![\frac{0.00600 M/s}{0.144 M/s}=\frac{k[0.0100 M]^x\times [0.0100 M]^1}{k[0.0200 M]^x\times [0.0300 M]^1}](/tpl/images/0515/4935/e9761.png)
![0.00600 M/s=k[0.0100 M]^3[0.0100 M]^1](/tpl/images/0515/4935/2cbdd.png) ..[1]
..[1]![k=\frac{0.00600 M/s}{[0.0100 M]^3[0.0100 M]^1}=6\times 10^5 M^{-3}s^{-1}](/tpl/images/0515/4935/88d8d.png)


