Chemistry, 19.02.2020 20:12 dirgoperadiego2122
The following information is given for benzene, C6H6, at 1atm: boiling point = 80.1 °C Hvap(80.1 °C) = 30.7 kJ/mol specific heat liquid = 1.74 J/g°C At a pressure of 1 atm, what is H in kJ for the process of condensing a 24.8 g sample of gaseous benzene at its normal boiling point of 80.1 °C. kJ

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle i need : ( asap i go it never mind
Answers: 2



Chemistry, 22.06.2019 23:00
Which type of intermolecular attractions holds ammonia molecules together with other ammonia molecules?
Answers: 3
You know the right answer?
The following information is given for benzene, C6H6, at 1atm: boiling point = 80.1 °C Hvap(80.1 °C)...
Questions




Mathematics, 28.08.2019 21:00




Mathematics, 28.08.2019 21:00

English, 28.08.2019 21:00





Biology, 28.08.2019 21:00


English, 28.08.2019 21:00

Arts, 28.08.2019 21:00


Mathematics, 28.08.2019 21:00

Mathematics, 28.08.2019 21:00

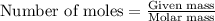
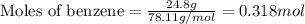
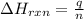
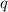 = amount of heat absorbed = ?
= amount of heat absorbed = ?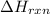 = enthalpy change of the reaction = 30.7 kJ/mol
= enthalpy change of the reaction = 30.7 kJ/mol