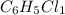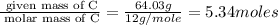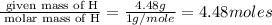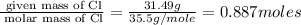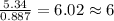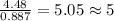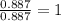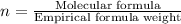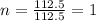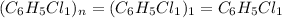Chemistry, 19.02.2020 23:18 zuleromanos
The reaction of equal molar amounts of benzene, C6H6, and chlorine, Cl2, carried out under special conditions, yields a gas and a clear liquid. Analysis of the liquid shows that it contains 64.03% carbon, 4.48% hydrogen, and 31.49% chlorine by mass and that is has a molar mass of 112.5 g/mol. The molecular formula will be determined. First, determine the number of moles of carbon in a 100 g sample.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

You know the right answer?
The reaction of equal molar amounts of benzene, C6H6, and chlorine, Cl2, carried out under special c...
Questions



History, 23.07.2019 10:50


Arts, 23.07.2019 10:50

History, 23.07.2019 10:50



History, 23.07.2019 10:50


Biology, 23.07.2019 10:50

Biology, 23.07.2019 10:50




Computers and Technology, 23.07.2019 10:50

Chemistry, 23.07.2019 10:50



Mathematics, 23.07.2019 10:50