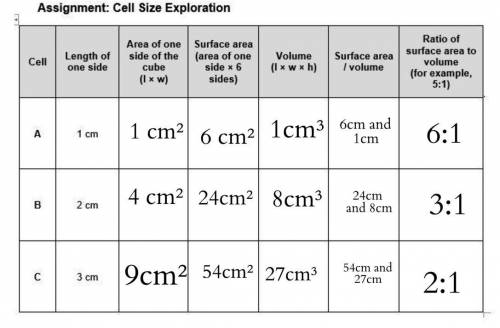
All the squares outlined on the model cells are 1 cm by 1 cm.
.Each face of Cube A measu...

All the squares outlined on the model cells are 1 cm by 1 cm.
.Each face of Cube A measures 1 cm long by 1 cm wide.
.Each face of Cube B measures 2 cm long by 2 cm wide.
.Each face of Cube C measures 3 cm long by 3 cm wide.
.Calculate the area of one side of each cube by multiplying the length times the width.
.Calculate the surface area of each cube by multiplying the area of one side of the cube by 6 sides.
.Calculate the volume of each cube by multiplying length times width times height.
.Build the ratio for each cube: (surface area)/volume
.For each cube, divide surface area by volume and round off to the nearest whole number. For example, 1.5 to 1 is 3:2.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1

Chemistry, 22.06.2019 17:30
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1

Chemistry, 23.06.2019 03:30
Ineed pls urgent 1-20 in order and fully detail step my step.
Answers: 1
You know the right answer?
Questions

Spanish, 29.10.2020 19:50

Health, 29.10.2020 19:50

Arts, 29.10.2020 19:50



Mathematics, 29.10.2020 19:50


Social Studies, 29.10.2020 19:50

Mathematics, 29.10.2020 19:50

History, 29.10.2020 19:50


English, 29.10.2020 19:50



Mathematics, 29.10.2020 19:50