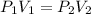Chemistry, 20.02.2020 22:05 luludawn3874
In response to Boyle’s Law, the pressure of a gas increases as the volume decreases because
the gas particles strike the walls of the container more often
the temperature of the gas increases
the gas particles get bigger
the gas particles strike the walls of the container with more force

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
In response to Boyle’s Law, the pressure of a gas increases as the volume decreases because
Questions




Mathematics, 24.03.2020 21:28


History, 24.03.2020 21:28





Computers and Technology, 24.03.2020 21:28


History, 24.03.2020 21:28


Social Studies, 24.03.2020 21:29




Mathematics, 24.03.2020 21:29

Mathematics, 24.03.2020 21:29