
Chemistry, 21.02.2020 02:27 nayelimoormann
Consider the following equilibrium process at 686°C. CO2(g) + H2(g) equilibrium reaction arrow CO(g) + H2O(g) The equilibrium concentrations of the reacting species are [CO] = 0.050 M, [H2] = 0.045 M, [CO2] = 0.086 M, and [H2O] = 0.040 M. (a) Calculate Kc for the reaction at 686°C. WebAssign will check your answer for the correct number of significant figures. (b) If we add CO2 to increase its concentration to 0.50 mol/L, what will the concentrations of all the gases be when equilibrium is reestablished?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 3

Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1
You know the right answer?
Consider the following equilibrium process at 686°C. CO2(g) + H2(g) equilibrium reaction arrow CO(g)...
Questions

History, 01.10.2019 07:50



Mathematics, 01.10.2019 07:50






History, 01.10.2019 07:50



Mathematics, 01.10.2019 07:50


Mathematics, 01.10.2019 07:50





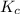 is 0.52.
is 0.52.![[CO_2] = 0.4748 M](/tpl/images/0518/6100/1098f.png)
![[H_2] = 0.0198 M](/tpl/images/0518/6100/4696c.png)
![[CO] = 0.0752 M](/tpl/images/0518/6100/1d075.png)
![[H_2O] =0.0652 M](/tpl/images/0518/6100/30296.png)
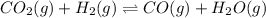
![[CO] = 0.050 M, [H_2] = 0.045 M, [CO_2] = 0.086 M, and [H_2O] = 0.040 M](/tpl/images/0518/6100/10b47.png)
![\K_c=\frac{[CO][H_2O]}{[CO_2][H_2]}](/tpl/images/0518/6100/2fcaf.png)
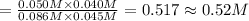
![K_c=\frac{[CO][H_2O]}{[CO_2][H_2]}](/tpl/images/0518/6100/c597d.png)
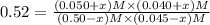
![[CO_2] = (0.50-x) M=(0.50-0.0252)M = 0.4748 M](/tpl/images/0518/6100/7357e.png)
![[H_2] = (0.045-x) M= (0.045-0.0252) M=0.0198 M](/tpl/images/0518/6100/31260.png)
![[CO] = (0.050+x) M=(0.050+0.0252)M = 0.0752 M](/tpl/images/0518/6100/0b9e4.png)
![[H_2O] = (0.040+x) M=(0.040+0.0252) M=0.0652 M](/tpl/images/0518/6100/b7e5e.png)



