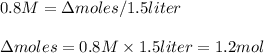Chemistry, 22.02.2020 18:57 kaleahearly123
Consider the following reaction:
2H2O2(aq)→2H2O(l)+O2(g)
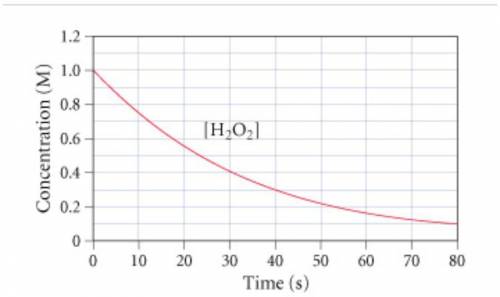
The graph (Figure 1) shows the concentration of H2O2 as a function of time.
If the instantaneous rate of formation of O2 is 3.3*(10^-3) moles/(liters*seconds), then...
If the initial volume of the H2O2 solution is 1.5 L , what total amount of O2 (in moles) is formed in the first 50 s of reaction?
Express your answer using two significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1


You know the right answer?
Consider the following reaction:
2H2O2(aq)→2H2O(l)+O2(g)
The graph (Figure 1...
2H2O2(aq)→2H2O(l)+O2(g)
The graph (Figure 1...
Questions

Physics, 03.10.2019 08:50

Physics, 03.10.2019 08:50

History, 03.10.2019 08:50


Mathematics, 03.10.2019 08:50

Health, 03.10.2019 08:50

Biology, 03.10.2019 08:50

Health, 03.10.2019 08:50


English, 03.10.2019 08:50

Computers and Technology, 03.10.2019 08:50





Biology, 03.10.2019 08:50

History, 03.10.2019 08:50


Social Studies, 03.10.2019 08:50

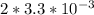
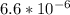
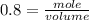
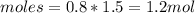
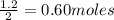 O₂ is formed in the first 50 s of reaction.
O₂ is formed in the first 50 s of reaction.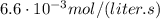
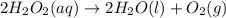
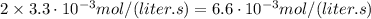
![\Delta [H_2O_2]=\Delta moles/Volume(liters)](/tpl/images/0520/3300/3d5f9.png)