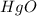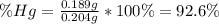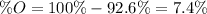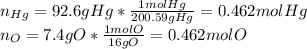Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
An oxide of mercury will thermally decompose when heated. A 0.204 gram sample of the mercury oxide i...
Questions

English, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

English, 17.09.2020 02:01

History, 17.09.2020 02:01

German, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

Social Studies, 17.09.2020 02:01

Biology, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

Biology, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

History, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

English, 17.09.2020 02:01

Mathematics, 17.09.2020 02:01

Social Studies, 17.09.2020 02:01