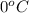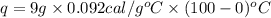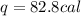Chemistry, 25.02.2020 04:30 hela9astrid
The specific heat capacity of copper is 0.092 calories per gram per degree Celsius. How much heat is required to raise the temperature of a 9 g piece of copper from 0°C to 100°C?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Type the correct answer in the box. spell all words correctly.what is the correct term for living the most sustainable life you can within your current circumstances? when your are being as sustainable as you can within your current lifestyle, you are said to be sustainability.
Answers: 3

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 04:00
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2
You know the right answer?
The specific heat capacity of copper is 0.092 calories per gram per degree Celsius. How much heat is...
Questions

Arts, 06.05.2020 01:33

Mathematics, 06.05.2020 01:33

Mathematics, 06.05.2020 01:33


History, 06.05.2020 01:33

Mathematics, 06.05.2020 01:33

History, 06.05.2020 01:33



Social Studies, 06.05.2020 01:33


Mathematics, 06.05.2020 01:33




World Languages, 06.05.2020 01:33




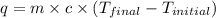
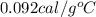
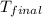 = final temperature =
= final temperature = 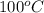
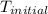 = initial temperature =
= initial temperature =