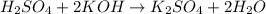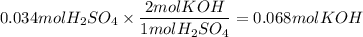The reaction of sulfuric acid (H2SO4) with potassium hydroxide (KOH) is described by the equation below. Suppose
0.06 L of KOH with unknown concentration is placed in a flask with bromthymol blue indicator. A solution of 0.20 M
H2504 is dripped into the KOH solution. After exactly 0.017 L of H2SO4 is added, the Indicator changes from blue to
yellow. What is the concentration of the KOH? You must show all of your work to earn credit. (4 points)
H2SO4 + 2KOH → K2SO4 + 2H20

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 21:00
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1
You know the right answer?
The reaction of sulfuric acid (H2SO4) with potassium hydroxide (KOH) is described by the equation be...
Questions

Mathematics, 28.05.2020 23:00

History, 28.05.2020 23:00

Mathematics, 28.05.2020 23:00

History, 28.05.2020 23:00


Physics, 28.05.2020 23:00

Mathematics, 28.05.2020 23:00





Mathematics, 28.05.2020 23:00

History, 28.05.2020 23:00


Mathematics, 28.05.2020 23:00



Chemistry, 28.05.2020 23:00

History, 28.05.2020 23:00