
The reaction below is carried out at a different temperature at which Kc=0.055. This time, however, the reaction mixture starts with only the product, [NO]=0.0100M, and no reactants. Find the equilibrium concentrations of N2, O2, and NO at equilibrium. N2(g)+O2(g)⇌2NO(g)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
If 200.0g of copper(ll) sulfate react with an excess of zinc metal, what is the theoretical yield of copper
Answers: 1

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3

Chemistry, 22.06.2019 23:00
What is the formula of the ionic compound composed of calcium cations and chloride anions
Answers: 1
You know the right answer?
The reaction below is carried out at a different temperature at which Kc=0.055. This time, however,...
Questions



Mathematics, 28.06.2019 15:00

Mathematics, 28.06.2019 15:00

Mathematics, 28.06.2019 15:00


History, 28.06.2019 15:00





History, 28.06.2019 15:00

History, 28.06.2019 15:00

Biology, 28.06.2019 15:00



History, 28.06.2019 15:00



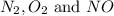 at equilibrium is, 0.0045 M, 0.0045 M and 0.001 M respectively.
at equilibrium is, 0.0045 M, 0.0045 M and 0.001 M respectively.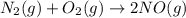
![K=\frac{[NO]^2}{[N_2][O_2]}](/tpl/images/0523/5256/ef70d.png)
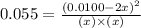
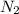 at equilibrium = x = 0.0045 M
at equilibrium = x = 0.0045 M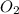 at equilibrium = x = 0.0045 M
at equilibrium = x = 0.0045 M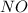 at equilibrium = (0.0100-2x) = (0.0100-2(0.0045)) = 0.001 M
at equilibrium = (0.0100-2x) = (0.0100-2(0.0045)) = 0.001 M

