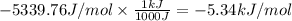PART A: Use the following glycolytic reaction to answer the question. If the concentration of DHAP is 0.125 M and the concentration of G3P is 0.06 M in a cell, what is the free energy change (ΔG)? Give your answer in 3 significant figures. (NOTE: Units are asked for in Part B.)
DHAP -> G3P (reversible)
Keq=5.4e-2
PART B: Units are important for all mathematical calculations. What are the units for free energy change in Part A?
PART C: Select ALL answers that correctly complete this sentence. Based on the calculated free energy change in Part A, the
a) forward reaction is favorable
b)forward reaction is unfavorable
c) reverse reaction is favorable
d) reverse reaction is unfavorable.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Complete this brønsted-lowry reaction placing each product by its appropriate label. hso4- + hcn
Answers: 1

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 12:40
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1
You know the right answer?
PART A: Use the following glycolytic reaction to answer the question. If the concentration of DHAP i...
Questions


Mathematics, 19.07.2019 06:30


Social Studies, 19.07.2019 06:30



Chemistry, 19.07.2019 06:30


Social Studies, 19.07.2019 06:30




Social Studies, 19.07.2019 06:30

Biology, 19.07.2019 06:30

Mathematics, 19.07.2019 06:30




Chemistry, 19.07.2019 06:30

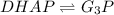
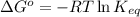
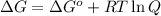
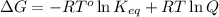
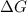 = Free energy change
= Free energy change 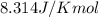
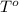 = standard temperature =
= standard temperature = ![25^oC=[273+25]K=298K](/tpl/images/0523/9218/0e82f.png)
![37^oC=[273+37]K=310K](/tpl/images/0523/9218/c6b28.png)
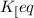 = equilibrium constant =
= equilibrium constant = 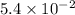
![\frac{[G_3P]}{[DHAP]}](/tpl/images/0523/9218/12d19.png)
![[G_3P]](/tpl/images/0523/9218/572f0.png) = 0.06 M
= 0.06 M![\Delta G=[-(8.314J/mol.K\times 298K\times \ln (5.4\times 10^{-2}))]+[(8.314J/mol.K\times 310K\times \ln (\frac{0.06}{0.125}))]\\\\\Delta G=-[-7231.46]+[-1891.7]=-5339.76J/mol](/tpl/images/0523/9218/63161.png)