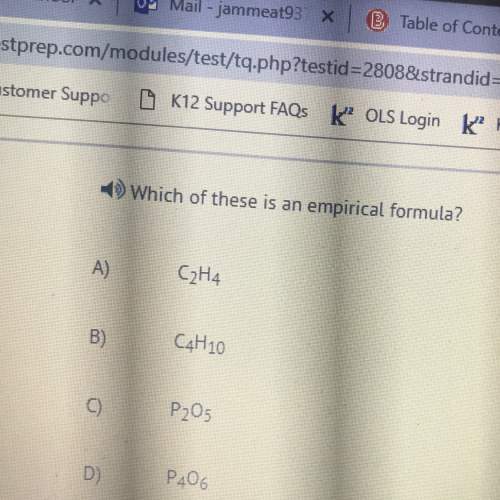
In the reaction, A → Products, the rate constant is 7.5 × 10−3 M−1 s−1.
If the initial...

In the reaction, A → Products, the rate constant is 7.5 × 10−3 M−1 s−1.
If the initial concentration of A is 0.613 M, what will be the concentration of A (in M) at t = 11.5 s?
Only enter the numerical value with three significant figures in the answer box below. Do NOT type in the unit (M).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1


Chemistry, 22.06.2019 16:00
Uranium can supply energy for the worlds electricity without admitting harmful greenhouse gases which of these statements best describes an outcome of uranium mining
Answers: 1

Chemistry, 22.06.2019 17:00
Complete each row of the table below by filling in the missing prefix or missing exponent.
Answers: 1
You know the right answer?
Questions



Mathematics, 19.09.2019 21:00


Chemistry, 19.09.2019 21:00



Social Studies, 19.09.2019 21:00

Biology, 19.09.2019 21:00

Mathematics, 19.09.2019 21:00

Mathematics, 19.09.2019 21:00



Biology, 19.09.2019 21:00


Physics, 19.09.2019 21:00

Computers and Technology, 19.09.2019 21:00



Physics, 19.09.2019 21:00