
Chemistry, 26.02.2020 01:54 xxtonixwilsonxx
Consider the following system at equilibrium:A(aq)+B(aq) <---> 2C(aq)Classify each of the following actions by whether it causes a leftward shift, a rightward shift, or no shift in the direction of the net reaction.1. Increase A- Right2. Increase B- Right3. Increase C- Left4. Decrease A- Right5. Decrease B- Right6. Decrease C- lEft7. Double A and Halve B- NO Shift8. Double both B and C- NO shift

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Which term best describes the form sound takes as it travels away from a drum (a- gas)(b-music) ( c-waves) (d-particles
Answers: 3

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1
You know the right answer?
Consider the following system at equilibrium:A(aq)+B(aq) <---> 2C(aq)Classify each of the foll...
Questions


Biology, 25.11.2021 05:20

Mathematics, 25.11.2021 05:20


Mathematics, 25.11.2021 05:20




German, 25.11.2021 05:20

Business, 25.11.2021 05:20


History, 25.11.2021 05:20


Social Studies, 25.11.2021 05:20



History, 25.11.2021 05:20



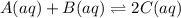
![K=\frac{[C]^2}{[A][B]}](/tpl/images/0524/2481/477c9.png)
![K'=\frac{[C]^2}{[2A][\frac{B}{2}]}=\frac{[C]^2}{[A][B]}](/tpl/images/0524/2481/7596a.png)
![K'=\frac{[2C]^2}{[A][2B]}=\frac{4[C]^2}{[A][2B]}=\frac{2[C]^2}{[A][B]}](/tpl/images/0524/2481/c241a.png)



