
Chemistry, 26.02.2020 17:02 coleman4405
According to the following balanced reaction, how many moles of KO are required to exactly react with 4.33 moles of H2O?4 KO(s) + 2 H2O(l) → 4 KOH(s) + O2(g)

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
You know the right answer?
According to the following balanced reaction, how many moles of KO are required to exactly react wit...
Questions

Mathematics, 20.08.2019 09:50


Social Studies, 20.08.2019 09:50

History, 20.08.2019 09:50


History, 20.08.2019 09:50


Mathematics, 20.08.2019 09:50





Mathematics, 20.08.2019 09:50


Geography, 20.08.2019 09:50


History, 20.08.2019 09:50

Mathematics, 20.08.2019 09:50


History, 20.08.2019 09:50

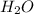 = 4.33 mol
= 4.33 mol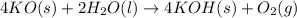
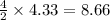 moles of KO
moles of KO

