
Chemistry, 26.02.2020 17:01 vanessasantos2004vs
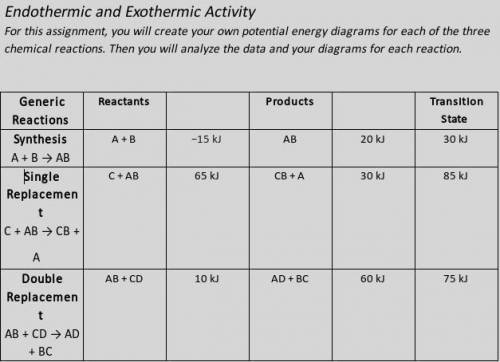
Illustrate the x- and y-axes to show the reaction pathway and potential energy, in kilojoules. Ensure your energy intervals are appropriate for the data.
Plot the enthalpy values of the reactants, products, and transition state using three horizontal dotted lines across the graph for each.
Draw the energy curve from the reactants line to the transition state and curve the line back down to the energy of the products. Label the reactants, products, and transition state.
Illustrate double-headed arrows to represent both the total change in enthalpy (ΔH) and the activation energy (Ea).
Calculate the total change in enthalpy and the activation energy using the energy values provided for each reaction. Record those values below the graph.
Make sure correct units are included.
I know this is a lot, so I'm giving more points than I ever have. Thank you!!!

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1


Chemistry, 23.06.2019 00:00
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3
You know the right answer?
Illustrate the x- and y-axes to show the reaction pathway and potential energy, in kilojoules. Ensur...
Questions





Chemistry, 08.07.2019 18:40



Computers and Technology, 08.07.2019 18:40

Mathematics, 08.07.2019 18:40

Computers and Technology, 08.07.2019 18:40


History, 08.07.2019 18:40

Social Studies, 08.07.2019 18:40







History, 08.07.2019 18:40



