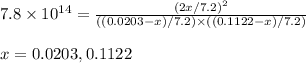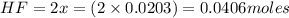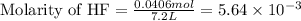Chemistry, 26.02.2020 19:52 krystalruby1219
Consider the formation of hydrogen fluoride: H2(g) + F2(g) ↔ 2HF(g) If a 3.9 L nickel reaction container (glass cannot be used because it reacts with HF) filled with 0.0052 M H2 is connected to a 3.3 L container filled with 0.034 M F2. The equilibrium constant, Kp, is 7.8 x 1014 (Hint, this is a very large number, what does that imply?) Calculate the molar concentration of HF at equilibrium. A further hint is provided after the first attempt in the feedback.

Answers: 1


Another question on Chemistry


Chemistry, 21.06.2019 19:30
If the element whose electric configuration ends in the d sublevel, the element is calssified as? a.inner transition b.noble gases c.representative d. transition
Answers: 2

Chemistry, 22.06.2019 01:00
Consider three unlabeled bottles, each contain small pieces of one of the following metals. - magnesium - sodium - silver the following reagents are used for identifying the metals. - pure water - a solution of 1.0 molar hcl - a solution of concentrated hno3 (a) which metal can be easily identified because it is much softer than the other two? describe a chemical test that distinguishes this metal from the other two, using only one of the reagents above. write a balanced chemical equation for the reaction that occurs. (b) one of the other two metals reacts readily with the hcl solution. identify the metal and write the balanced chemical equation for the reaction that occurs when this metal is added to the hcl solution. use the table of standard reduction potentials (attached) to account for the fact that this metal reacts with hcl while the other does not. (c) the one remaining metal reacts with the concentrated hno3 solution. write a balanced chemical equation for the reaction that occurs. (d) the solution obtained in (c) is diluted and a few drops of 1 m hcl is added. describe what would be observed. write a balanced chemical equation for the reaction that occurs.
Answers: 2

You know the right answer?
Consider the formation of hydrogen fluoride: H2(g) + F2(g) ↔ 2HF(g) If a 3.9 L nickel reaction conta...
Questions

English, 06.08.2021 04:50





Engineering, 06.08.2021 04:50

English, 06.08.2021 04:50


Chemistry, 06.08.2021 04:50


Mathematics, 06.08.2021 04:50


English, 06.08.2021 04:50


English, 06.08.2021 04:50

Mathematics, 06.08.2021 04:50

Mathematics, 06.08.2021 04:50

Chemistry, 06.08.2021 04:50


History, 06.08.2021 04:50

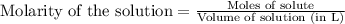 ......(1)
......(1)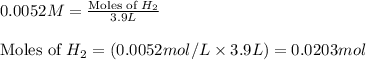
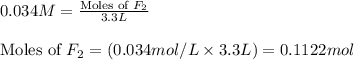
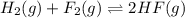
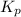 with
with 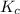 is given by the formula:
is given by the formula: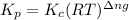
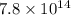
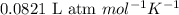
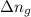 = change in number of moles of gas particles =
= change in number of moles of gas particles = 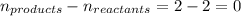
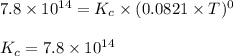
![K_c=\frac{[HF]^2}{[F_2][H_2]}](/tpl/images/0525/2814/b6e65.png)
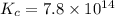
![[HF]_{eq}=\frac{2x}{7.2}](/tpl/images/0525/2814/4c924.png)
![[H_2]_{eq}=\frac{(0.0203)}{7.2}](/tpl/images/0525/2814/1a13d.png)
![[F_2]_{eq}=\frac{(0.1122-x)}{7.2}](/tpl/images/0525/2814/7c73d.png)