
Chemistry, 27.02.2020 00:58 aredwolf2017
Enter your answer in the provided box. Sodium stearate (C17H35COONa) is a major component of bar soap. The Ka of the stearic acid is 1.3 × 10−5. What is the pH of 10.0 mL of a solution containing 3.96 g of sodium stearate?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The minerals found in bones are deposited by living cells called
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3


Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
You know the right answer?
Enter your answer in the provided box. Sodium stearate (C17H35COONa) is a major component of bar soa...
Questions

Mathematics, 07.12.2021 02:50

History, 07.12.2021 02:50

Biology, 07.12.2021 02:50





Mathematics, 07.12.2021 02:50

English, 07.12.2021 02:50

Social Studies, 07.12.2021 02:50

Biology, 07.12.2021 02:50

Physics, 07.12.2021 02:50

Mathematics, 07.12.2021 02:50




SAT, 07.12.2021 02:50


Chemistry, 07.12.2021 02:50

Chemistry, 07.12.2021 02:50

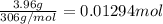
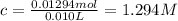
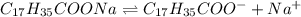
![[C_{17}H_{35}COO^-]=c=1.294 M](/tpl/images/0525/9707/45651.png)
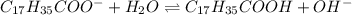
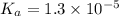
![K_a=\frac{[C_{17}H_{35}COOH][OH^-]}{[C_{17}H_{35}COO^-]}](/tpl/images/0525/9707/6f054.png)
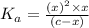
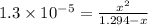
![[OH^-]=0.0041 M](/tpl/images/0525/9707/2d6f6.png)
![pOH=-\log[OH^-]=-\log[0.0041 M]=2.39](/tpl/images/0525/9707/63803.png)


