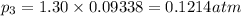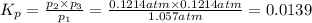Chemistry, 27.02.2020 02:54 MacieKay8865
At 25°C, gaseous decomposes to and to the extent that 10.3% of the original (by moles) has decomposed to reach equilibrium. The total pressure (at equilibrium) is 1.30 atm. Calculate the value of for this system. =

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

You know the right answer?
At 25°C, gaseous decomposes to and to the extent that 10.3% of the original (by moles) has decompose...
Questions





Social Studies, 12.12.2019 02:31




Biology, 12.12.2019 02:31



Mathematics, 12.12.2019 02:31


Health, 12.12.2019 02:31


Biology, 12.12.2019 02:31




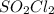 decomposes to
decomposes to 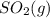 and
and 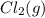 to the extent that 10.3% of the original
to the extent that 10.3% of the original 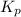 of the reaction is 0.0139.
of the reaction is 0.0139.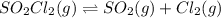
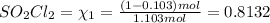
 (Dalton's law of partial pressure)
(Dalton's law of partial pressure)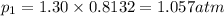
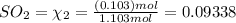
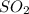 equilibrium ;
equilibrium ;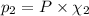 (Dalton's law of partial pressure)
(Dalton's law of partial pressure)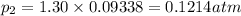
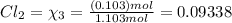
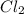 equilibrium ;
equilibrium ;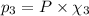 (Dalton's law of partial pressure)
(Dalton's law of partial pressure)