
Chemistry, 27.02.2020 03:08 gabrielbergemancat
Urea (CH4N2O) is a common fertilizer that can be synthesized by the reaction of ammonia (NH3) with carbon dioxide as follows: 2NH3(aq)+CO2(aq)→CH4N2O(aq)+H2O(l) In an industrial synthesis of urea, a chemist combines 135.9 kg of ammonia with 211.4 kg of carbon dioxide and obtains 178.0 kg of urea.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is the cellular process that releases the energy stored in food molecules
Answers: 3

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2
You know the right answer?
Urea (CH4N2O) is a common fertilizer that can be synthesized by the reaction of ammonia (NH3) with c...
Questions

Physics, 29.01.2021 18:50


English, 29.01.2021 18:50

Advanced Placement (AP), 29.01.2021 18:50

Mathematics, 29.01.2021 18:50

Mathematics, 29.01.2021 18:50


Arts, 29.01.2021 18:50



Mathematics, 29.01.2021 18:50

Physics, 29.01.2021 18:50


Mathematics, 29.01.2021 18:50

English, 29.01.2021 18:50

Mathematics, 29.01.2021 18:50

Mathematics, 29.01.2021 18:50

Mathematics, 29.01.2021 18:50

Arts, 29.01.2021 18:50

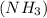
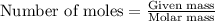 .....(1)
.....(1)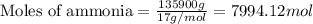
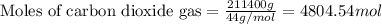
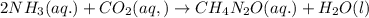
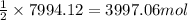 of carbon dioxide
of carbon dioxide


