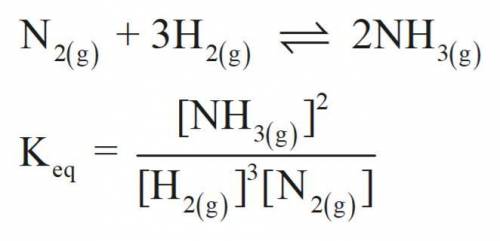Chemistry, 27.02.2020 19:26 Batzs3rdacct
Consider the following chemical equilibrium:
N2(g) + 3H2(g) ⇌ 2NH3
Now write an equation below that shows how to calculate from for this reaction at an absolute temperature . You can assume is comfortably above room temperature.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2


Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1
You know the right answer?
Consider the following chemical equilibrium:
N2(g) + 3H2(g) ⇌ 2NH3
Now writ...
N2(g) + 3H2(g) ⇌ 2NH3
Now writ...
Questions

Mathematics, 18.03.2021 03:10



Mathematics, 18.03.2021 03:10


Mathematics, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10




Mathematics, 18.03.2021 03:10

Geography, 18.03.2021 03:10






Mathematics, 18.03.2021 03:10

History, 18.03.2021 03:10