Chemistry, 28.02.2020 04:38 shelbylynn17
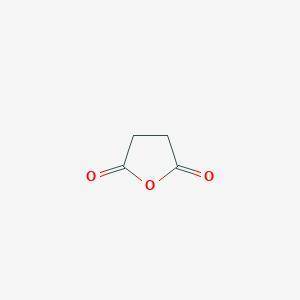
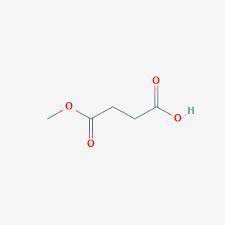
A compound A has prominent infrared absorptions at 1050, 1786, and 1852 cm–1 and shows a single absorption in the proton NMR spectrum at δ 3.00. When heated gently with methanol, compound B, C5H8O4, is obtained. Compound B has IR absorptions at 2500–3000 (broad), 1730, and 1701 cm–1, and its proton NMR spectrum in D2O consists of resonances at δ 2.7 (complex splitting) and δ 3.7 (a singlet) in the intensity ratio 4:3. Give the structures A and B, omitting stereochemistry.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1


Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1
You know the right answer?
A compound A has prominent infrared absorptions at 1050, 1786, and 1852 cm–1 and shows a single abso...
Questions

Mathematics, 12.07.2019 10:30

Chemistry, 12.07.2019 10:30


Mathematics, 12.07.2019 10:30

English, 12.07.2019 10:30

Mathematics, 12.07.2019 10:30

Mathematics, 12.07.2019 10:30




Mathematics, 12.07.2019 10:30

Spanish, 12.07.2019 10:30

Mathematics, 12.07.2019 10:30


Mathematics, 12.07.2019 10:30


Mathematics, 12.07.2019 10:30

Mathematics, 12.07.2019 10:30

Mathematics, 12.07.2019 10:30