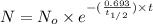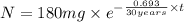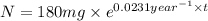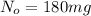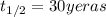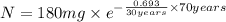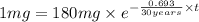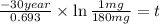Chemistry, 28.02.2020 19:22 gianababnnna
The half-life of cesium-137 is 30 years. Suppose we have a 180 mg sample. Find the mass that remains after t years. Let y(t) be the mass (in mg) remaining after t years. Then we know the following. y ( t ) = y ( 0 ) e k t = ⋅ e k t Since the half-life is 30 years, then y ( 30 ) = mg. Thus, e 30 k = . Therefore, k = . Now, remembering that ln x n = n ln x and that e ln z = z , we have y ( t ) = 180 e ( t / 30 ) ( − ln 2 ) = mg. How much of the sample remains after 70 years? After 70 years we have the following. y ( 70 ) = 180 ⋅ 2 = mg (Round your answer to two decimal places.) After how long will only 1 mg remain? To find the time at which only 1 mg remains, we must solve 1 = y ( t ) = 180 ( 2 − t / 30 ) , and so we get the following. t = − 30 log 2 ( ) Hence, we conclude the following. t = yr (Round your final answer to one decimal place.)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Review the branily terms and services guides well u know what i never did so go have a nice ice cream sunday
Answers: 1

Chemistry, 22.06.2019 02:30
At 40 âc the solution has at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.g of kno3 per 100 g of water and it can contain up to at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.g of kno3 per 100 g of water. at 0 âc the solubility is ~ at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.kno3 per 100 g of water, so ~ at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.gkno3 per 100 g of water will precipitate out of solution. a kno3 solution containing 55 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 2

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1
You know the right answer?
The half-life of cesium-137 is 30 years. Suppose we have a 180 mg sample. Find the mass that remains...
Questions




Mathematics, 14.10.2020 01:01


Mathematics, 14.10.2020 01:01




Mathematics, 14.10.2020 01:01


Advanced Placement (AP), 14.10.2020 01:01

English, 14.10.2020 01:01




Mathematics, 14.10.2020 01:01

Geography, 14.10.2020 01:01


History, 14.10.2020 01:01

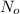 = 180 mg
= 180 mg
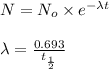
![t_{1/2}=30 years[p/tex]where, [tex]N_o](/tpl/images/0528/1653/35331.png) = initial mass of isotope
= initial mass of isotope
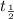 = half life of the isotope
= half life of the isotope
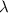 = rate constant
= rate constant