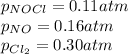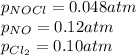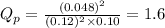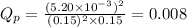Chemistry, 29.02.2020 00:00 KENYONWASHINGTON868
Nitric oxide (NO) reacts readily with chlorine gas as follows.2 NO(g) + Cl2(g) equilibrium reaction arrow 2 NOCl(g)At 700. K the equilibrium constant Kp for this reaction is 0.26. Predict the behavior of each of the following mixtures at this temperature and indicate whether or not the mixtures are at equilibrium. If not, state whether the mixture will need to produce more products or reactants to reach equilibrium.(a) PNO = 0.16 atm, PCl2 = 0.30 atm, and PNOCl = 0.11 atm.(b) PNO = 0.12 atm, PCl2 = 0.10 atm, and PNOCl = 0.048 atm.(c) PNO = 0.15 atm, PCl2 = 0.15 atm, and PNOCl = 5.20 10-3 atm.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1



Chemistry, 23.06.2019 01:00
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2
You know the right answer?
Nitric oxide (NO) reacts readily with chlorine gas as follows.2 NO(g) + Cl2(g) equilibrium reaction...
Questions

Mathematics, 25.06.2021 02:50


Mathematics, 25.06.2021 02:50


Chemistry, 25.06.2021 02:50


Mathematics, 25.06.2021 02:50

Mathematics, 25.06.2021 03:00

English, 25.06.2021 03:00



History, 25.06.2021 03:00


Mathematics, 25.06.2021 03:00

Mathematics, 25.06.2021 03:00




Computers and Technology, 25.06.2021 03:00

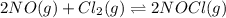
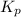 for above equation follows:
for above equation follows: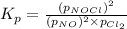 .....(1)
.....(1)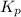 = 0.26
= 0.26 ; the reaction is product favored.When
; the reaction is product favored.When 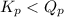 ; the reaction is reactant favored.When
; the reaction is reactant favored.When 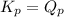 ; the reaction is in equilibrium.
; the reaction is in equilibrium.