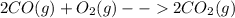Chemistry, 29.02.2020 01:40 ShlomoShekelstein
Consider the following hypothetical molecular collisions and predict which of the following will form potential products, given the values for the energy of activation, Ea, and enthalpy of the reaction, ΔH, in combination with the molecular orientation.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1

Chemistry, 22.06.2019 03:30
In saturated organic compounds, all the bonds between carbon atoms are called?
Answers: 1

Chemistry, 22.06.2019 05:20
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3
You know the right answer?
Consider the following hypothetical molecular collisions and predict which of the following will for...
Questions

Mathematics, 23.02.2021 19:10

Mathematics, 23.02.2021 19:10

Mathematics, 23.02.2021 19:10


Social Studies, 23.02.2021 19:10




Mathematics, 23.02.2021 19:10

Mathematics, 23.02.2021 19:10

Mathematics, 23.02.2021 19:10

English, 23.02.2021 19:10





Mathematics, 23.02.2021 19:10

Mathematics, 23.02.2021 19:10


Chemistry, 23.02.2021 19:10