
Antimony pentachloride decomposes according to this equation: SbCl5(g)⇌SbCl3(g)+Cl2(g) An equilibrium mixture in a 5.00-L flask at 448 °C contains 3.85 g of SbCl5, 9.14 g of SbCl3, and 2.84 g of Cl2. How many grams of each will be found if the mixture is transferred into a 2.00-L flask at the same temperature?

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 02:00
Scientists are often interested in knowing the molar heat of combustion – the heat released during the combustion of one mole of a substance. use the periodic table to find molar masses. how many moles of ethanol are present in the sample?
Answers: 2

Chemistry, 23.06.2019 04:50
The diagin dilutepage 6 of 12a6a5(a)fluorine, chlorine, bromine and iodine are placed in the same group of theperiodic table.state the common name used to describe elements in this group.(i)state the group in which the elements are placed and explain whythey are placed in that group.(ii)which of the above named elements is a solid at roomtemperature and pressure?
Answers: 2


Chemistry, 23.06.2019 07:40
Which of the following has expanded our knowledge of the universe beyond our solar system the most? a. manned space travel b. the hubble space telescope c. the pioneer and voyager missions d. the international space station
Answers: 3
You know the right answer?
Antimony pentachloride decomposes according to this equation: SbCl5(g)⇌SbCl3(g)+Cl2(g) An equilibriu...
Questions




Mathematics, 18.10.2019 11:00

Physics, 18.10.2019 11:00




Computers and Technology, 18.10.2019 11:00

Advanced Placement (AP), 18.10.2019 11:00

Spanish, 18.10.2019 11:00



Mathematics, 18.10.2019 11:00



Mathematics, 18.10.2019 11:00

Mathematics, 18.10.2019 11:00


Mathematics, 18.10.2019 11:00

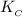 =
= ![\frac{[SbCl_3][Cl_2]}{[SbCl_5]}](/tpl/images/0528/9275/e005c.png)
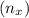 =
= 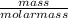
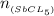 =
= 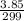
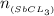 =
= 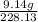
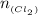 =
= 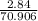
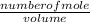
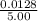
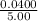
![\frac{[8*10^{-3}][8*10^{-3}]}{[2.56*10^{-3}]}](/tpl/images/0528/9275/199f0.png)
![\frac{[y][y]}{[y]}](/tpl/images/0528/9275/d2bce.png)


