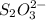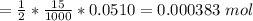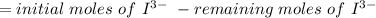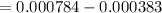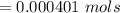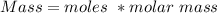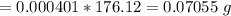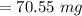Chemistry, 29.02.2020 02:54 kiarabermudez754
Calculate the weight percent of ascorbic acid in a tablet of Vitamin C from the following data:A 80 mg sample of a crushed Vitamin C tablet was dissolved in 40 mL of H2SO4 and 20 mL of water. Two grams of KI and 40. mL of 0.00653 M KIO3 solution was added, and the mixture titrated to a starch endpoint. The titration required 15 mL of 0.0510 M thiosulfate solution. I know the answer is 88%, but how did they get it?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Which object forms when a supergiant runs out of fuel? a red giant a black hole a white dwarf a neutron star
Answers: 1

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1
You know the right answer?
Calculate the weight percent of ascorbic acid in a tablet of Vitamin C from the following data:A 80...
Questions


Mathematics, 12.01.2021 17:40

Mathematics, 12.01.2021 17:40



Computers and Technology, 12.01.2021 17:40


Mathematics, 12.01.2021 17:40

Law, 12.01.2021 17:40

Computers and Technology, 12.01.2021 17:40







Computers and Technology, 12.01.2021 17:40


English, 12.01.2021 17:40

Advanced Placement (AP), 12.01.2021 17:40

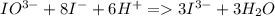
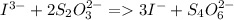
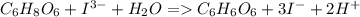
 = 3 × number of moles of
= 3 × number of moles of 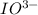
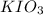
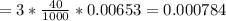 mol
mol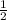 x moles of
x moles of