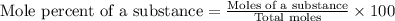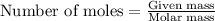Chemistry, 29.02.2020 05:37 valdezavery1373
A student obtains a mixture of the chlorides of two unknown metals, X and Z. The percent by mass of X and the percent by mass of Z in the mixture is known. Which of the following additional information is most helpful in calculating the mole percent of XCl(s) and of ZCl(s) in the mixture:.
a) the number of isotopes of Cl
b) the Molar masses of Z and X
c) The density of either XCL(s) or ZCl(s)
d) The eprcent by mass of Cl in the mixture

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 23:00
What prefix multiplier is appropriate for reporting a measurement of 5.57 ×10−5 m?
Answers: 1


Chemistry, 23.06.2019 03:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1
You know the right answer?
A student obtains a mixture of the chlorides of two unknown metals, X and Z. The percent by mass of...
Questions

Mathematics, 14.04.2021 21:00


Mathematics, 14.04.2021 21:10

English, 14.04.2021 21:10

Arts, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10


Mathematics, 14.04.2021 21:10



Arts, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10





Arts, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10