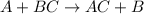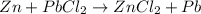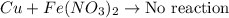Chemistry, 02.03.2020 17:03 ssargeant2559
Write a balanced chemical equation for each single replacement reaction that takes place in aqueous solution. write no reaction if a reaction does not occur

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 01:30
Select the correct answer from each drop-down menu. to make a table of the elements, dmitri mendeleev sorted the elements according to their . he then split the list of elements into several columns so that elements beside each other had similar .
Answers: 2
You know the right answer?
Write a balanced chemical equation for each single replacement reaction that takes place in aqueous...
Questions

Biology, 25.02.2021 18:20





Mathematics, 25.02.2021 18:20

History, 25.02.2021 18:20

English, 25.02.2021 18:20

Mathematics, 25.02.2021 18:20

Mathematics, 25.02.2021 18:20

English, 25.02.2021 18:20

Social Studies, 25.02.2021 18:20



Mathematics, 25.02.2021 18:20


Advanced Placement (AP), 25.02.2021 18:20

Mathematics, 25.02.2021 18:20