
Chemistry, 02.03.2020 18:29 mariahisawsome1
Manganese commonly occurs in nature as a mineral. The extraction of manganese from the carbonite mineral rhodochrosite, involves a two-step process. In the first step, manganese (II) carbonate and oxygen react to form manganese (IV) oxide and carbon dioxide: 2 MnCO3(s) + O2(g) → 2MnO2(s) + 2CO2(g) In the second step, manganese (IV) oxide and aluminum react to form manganese and aluminum oxide: 3 MnO2(s) + 4 Al(s) → 3 Mn(s) + 2 Al2O3(s) Write the net chemical equation for the production of manganese (II) carbonate, oxygen and aluminum. Be sure your equation is balanced.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 23.06.2019 01:30
Which statement accurately represents the arrangement of electrons in bohr’s atomic model?
Answers: 2

Chemistry, 23.06.2019 01:50
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1
You know the right answer?
Manganese commonly occurs in nature as a mineral. The extraction of manganese from the carbonite min...
Questions

Biology, 02.11.2019 12:31

Mathematics, 02.11.2019 12:31






Health, 02.11.2019 12:31


History, 02.11.2019 12:31


SAT, 02.11.2019 12:31

Mathematics, 02.11.2019 12:31


Chemistry, 02.11.2019 12:31


Mathematics, 02.11.2019 12:31

Advanced Placement (AP), 02.11.2019 12:31


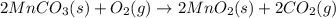
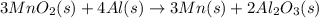
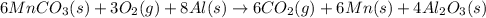
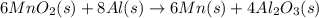
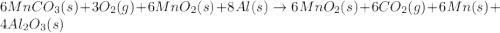
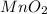 is common on both side, by cancelling it, we get:
is common on both side, by cancelling it, we get:

