
Consider the following three processes: (1) Melting of ice at room temperature (2) Boiling of water at 101°C (3) Dissolving of NH4NO3 with water in an instant cold pack? Which statement(s) that are true about ALL three of the given processes? (Choose one or more)(A) Endothermic(B) Exothermic(C) Nonspontaneous(D) Spontaneous(E) none of these statements can be used to describe all three processes

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1

Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1
You know the right answer?
Consider the following three processes: (1) Melting of ice at room temperature (2) Boiling of water...
Questions





Mathematics, 16.07.2019 19:00

Mathematics, 16.07.2019 19:00

Mathematics, 16.07.2019 19:00

Social Studies, 16.07.2019 19:00


Social Studies, 16.07.2019 19:00

Social Studies, 16.07.2019 19:00


Social Studies, 16.07.2019 19:00



Biology, 16.07.2019 19:00




Social Studies, 16.07.2019 19:00

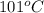 is also an endothermic process. This is because heat is absorbed by water molecules due to which their state changes from liquid to vapor form. When
is also an endothermic process. This is because heat is absorbed by water molecules due to which their state changes from liquid to vapor form. When 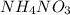 is dissolved in water then heat is absorbed as there occurs a decrease in temperature of water. Hence, it is also an endothermic reaction.
is dissolved in water then heat is absorbed as there occurs a decrease in temperature of water. Hence, it is also an endothermic reaction.

