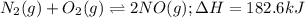Chemistry, 03.03.2020 05:02 emanihackle9
Nitrogen and oxygen react at high temperatures. N2(g) + O2(g) equilibrium reaction arrow 2 NO(g) ΔH = 182.6 kJ (a) What will happen to the concentrations of N2, O2, and NO at equilibrium if more O2 is added?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2

Chemistry, 23.06.2019 05:50
What are the coefficients to balance the following equation? ba+br=babr2
Answers: 1
You know the right answer?
Nitrogen and oxygen react at high temperatures. N2(g) + O2(g) equilibrium reaction arrow 2 NO(g) ΔH...
Questions



English, 26.11.2019 20:31


Social Studies, 26.11.2019 20:31

Mathematics, 26.11.2019 20:31





Biology, 26.11.2019 20:31

History, 26.11.2019 20:31

Computers and Technology, 26.11.2019 20:31



History, 26.11.2019 20:31



History, 26.11.2019 20:31

Mathematics, 26.11.2019 20:31