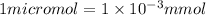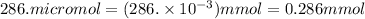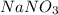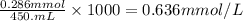Chemistry, 03.03.2020 05:49 nadiasifuentes15
A chemist prepares a solution of sodium nitrate (NaNO3) by measuring out 286. umol of sodium nitrate into a 450. mL volumetric flask and filling the flask to the mark with water. Calculate the concentration in mmol/L of the chemist's sodium nitrate solution.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 01:30
The biomedical technique in which a part of the brain is destroyed with electric current is known as a. electroconvulsive therapy b. prefrontal lobotomy c. bilateral cingulotomy d. tardive dyskinesia
Answers: 2
You know the right answer?
A chemist prepares a solution of sodium nitrate (NaNO3) by measuring out 286. umol of sodium nitrate...
Questions


Computers and Technology, 26.04.2020 05:03


Mathematics, 26.04.2020 05:03


Physics, 26.04.2020 05:03

History, 26.04.2020 05:03


History, 26.04.2020 05:03

Spanish, 26.04.2020 05:03

Advanced Placement (AP), 26.04.2020 05:03


History, 26.04.2020 05:04




Mathematics, 26.04.2020 05:04

Mathematics, 26.04.2020 05:04