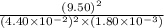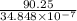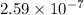Chemistry, 03.03.2020 06:14 alejandra216
Be sure to answer all parts. The equilibrium constant (Kc) for the formation of nitrosyl chloride, an orange-yellow compound, from nitric oxide and molecular chlorine 2NO(g) + Cl2(g) ⇌ 2NOCl(g) is 1 × 107 at a certain temperature. In an experiment, 4.40 × 10−2 mole of NO, 1.80 × 10−3 mole of Cl2, and 9.50 moles of NACl are mixed in a 2.60−L flask. What is Qc for the experiment

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

You know the right answer?
Be sure to answer all parts. The equilibrium constant (Kc) for the formation of nitrosyl chloride, a...
Questions



















Biology, 29.10.2019 05:31

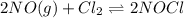
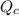 of this reaction is as follows.
of this reaction is as follows.![Q_{c} = \frac{[NOCl]^{2}}{[NO]^{2}[Cl_{2}]}](/tpl/images/0532/0245/90950.png)